|
Struthio camelus, the ostrich, is the largest member of the ratite birds, a group that includes the extant emu, rhea, kiwis, and cassowaries as well as the extinct moas and elephant birds. None of the species allocated to Ratitae are able to fly. Some, such as the ostrich and emu, are cursorial (runners); however, the kiwis and cassowaries live in densely forested areas. Although morphologically diverse, the ratites are connected by a number of synapomorphies including a fused coracoid and scapula, the trochlea of metatarsi II of the tarsometatarsus is not plantarly deflected and the distal end does not reach farther distally than the distal end of the trochlea of metatarsi IV, the musculus flexor hallucis longus to the hallux is either weak or absent, and the oil gland is only minutely tufted or naked (Mayr and Clarke, 2003). | |
The ratites belong to the more inclusive clade, Palaeognathae, a taxon that also includes the tinamous, a group of birds that unlike ratites is able to fly. Historically, the palaeognaths were grouped together based on the morphology of their palate (dromaeognathous or, more commonly referred to as the palaeognathous palate; Huxley, 1867; Pycraft, 1900). The features that comprise the palaeognathous palate are a broad vomer that unites rostrally with the broad maxillopalatine plates and caudally with both the palatines and the pterygiods, the lack of an articulation between the pterygoids and the parasphenoidal rostrum, basipterygoid processes that project from the body of the basisphenoid/parasphenoid complex rather than the parasphenoidal rostrum, the basisphenoidal processes articulate with the pterygoids near the distal ends of the pterygoids, and a single head to the quadrate (Huxley, 1867). In addition to these palatal characters, other morphological features also unite Palaeognathae: a marked furrow just rostral of the nasal opening on the upper beak, rostral extension of the mesethmoid beyond the naso-frontal hinge, two strong grooves on the ventral surface of the mandibular symphysis, a flat dorsal surface of the mandibular symphysis, three to four costal processes on the sternum, and a greatly reduced or no hallux (Mayr and Clarke, 2003). Monophyly of Palaeognathae, although historically disputed, is supported in recent morphological, molecular, and total evidence studies (Cracraft, 1974; Bledsoe, 1988, Härlid et al., 1997, 1998, 1999; Cooper et al., 2000; Livezey and Zusi, 2001, Mayr and Clarke, 2003).
The ostrich is restricted to northern Africa south of the Sahara, southwards to southwest Tanzania. One subspecies, S. camelus australis is isolated in southern Africa south of the Zambezi and Cunene Rivers. The modern geographical range of S. camelus is significantly contracted from its historical extension; ostriches once inhabited most of north and south Africa extending into the Arabian Peninsula (the last sighting in this region occurred as recently as the mid-20th century) (Davies, 2002).

About the Species
Specimen TMM M-4826 is a complete skeleton of a juvenile female Struthio camelus (exact age unknown), donated to the Texas Memorial Museum by the Abilene Zoo. The left vomerine process of the maxilla is broken. Because the specimen is a juvenile, the laterosphenoids are incompletely ossified along their anterior margins, and there are centers of incomplete ossification on either side of the midline of the supraoccipital.
Photos of the specimen itself (minus the lower jaw):
 |
 |
 |
|
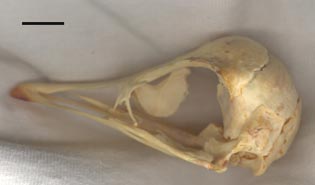
Left lateral view of the specimen. The scale bar is 1 cm. |
|
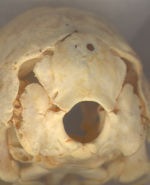
View of the occiput. |
 |
|
|
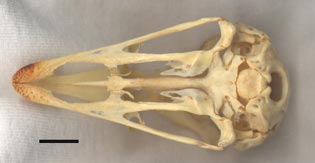
Ventral view of the specimen. The scale bar equals 1 cm. |
|
|

About this Specimen
This specimen was scanned by Matthew Colbert on 05 February 2001 at the University of Texas High-Resolution X-ray CT Facility. The slices were processed using the following software programs: Adobe Photoshop 6.0, Scion Image 4.0.2 Beta, and RenameMan 2.3. The images were first reduced from 16-bit format to 8-bit format using Photoshop. The images were then resliced in the horizontal and sagittal planes using Scion Image, after calculating the input slice spacing of 2.43 pixels (the image resolution [512 pixels] divided by the field of reconstruction [43mm] and then multiplied by the interslice spacing [0.204mm]). The output slice spacing of the resliced images was set to 1.00, resulting in an interslice spacing of 0.084 mm, with 429 horizontal slices and 410 sagittal slices (including one blank slice at the beginning and end of each data set). The image contrast was then adjusted for maximum contrast and minimum bone data loss. Adjusting the levels in Adobe Photoshop, the Input numbers used were 45/1/220, and the Output numbers were 0/1/239. Next, the slices in each plane were labeled with a number using the Scion Image macro that inserts the file name into the corner of each respective slice (the slices first had to be renamed using RenameMan to eliminate prefixes and file extensions, leaving only a three-digit number corresponding to the number of the slice).
After renumbering, 74 of the coronal slices and 30 of the horizontal slices were labeled using Adobe Illustrator 9.0. The bones were labeled in red (RBG value 255) using a standardized abbreviation in all caps (font: Myriad Roman 18); the lines indicating each bone were also in red with a stroke value of 2 pts. A maximum of five layers were used for each slice: the first was a locked image of the slice itself; the second was reserved for the bone labels; the third was reserved for labeling the processes; and the fourth was for sutures and other openings such as fenestrae and canals. Processes were labeled in yellow, with the same stroke and fill values as for the bones. Sutures were marked with a stroke value of 1 pt, and they and the other spaces were labeled in green . Finally, the wax used to affix the mandible and vertebrae to the cranium was marked in blue, with the same stroke and fill values as for the bones and processes. These labeled slices will be made available here once I finish my research.
This project was started in the Digital Methods for Paleontology course at The University of Texas at Austin. I'd like to thank Prof. Chris Bell and Brian Speer, DVM, for their assistance with reference material, as well as the members of my work group for their valuable input: Amy Balanoff, David Dufeau, Jonathan Franzosa, and Justin Hall. I'd also like to thank Matt Colbert, Ted Macrini, Jessie Maisano, and Farrah Welch for their troubleshooting tips. Funding for scanning was provided by a National Science Foundation Digital Libraries Initiative grant to Dr. Timothy Rowe of The University of Texas at Austin.

About the
Scan
Literature
Baumel, J.J. (ed.). 1993. Handbook of Avian Anatomy : Nomina Anatomica Avium (2nd ed.). Nuttall Ornithological Club, 779 pp.
Bell, C.J. and M. Colbert. 1999. Basic Vertebrate Cranial Osteology. CD-ROM - Data Test Version.
Bledsoe, A. H. 1988. A phylogenetic analysis of postcranial skeletal characters of the ratite birds. Annals of Carnegie Museum 57:73-90.
Cooper, A., C. Lalueza-Fox, S. Anderson, A. Rambaut, J. Austin, and R. Ward. 2000. Complete mitochondrial genome sequences of two extinct moas clarify ratite evolution. Nature 409:704-707.
Cracraft, J. 1974. Phylogeny and evolution of the ratite birds. Ibis 116:494-521.
Davies, S. J. J. F. 2002. Ratites and Tinamous. Oxford University Press, Oxford, 310 pp.
Hildebrand, M. 1995. Analysis of Vertebrate Structure (4th ed.). John Wiley & Sons, 657 pp.
Härlid, A., A. Janke, and U. Arnason. 1997. The mtDNA sequence of the ostrich and the divergence between paleognathous and neognathous birds. Molecular Biology and Evolution 14:754-761.
Härlid, A., A. Janke, and U. Arnason. 1998. The complete mitochondrial genome of Rhea americana and early avian divergences. Journal of Molecular Evolution 46:669-679.
Härlid, A., A. Janke, and U. Arnason. 1999. Analyses of mitochondrial DNA nest ratite birds within the Neognathae: supporting a neotenous origin of ratite morphological characters. Proceedings of the Royal Society of London, B, 266:305-309.
Huxley, T. H. 1867. On the classification of birds; and on the taxonomic value of the modifications of certain of the cranial bones observable in that class. Proceedings of the Zoological Society of London 1867:415-472.
Livezey, B. C., and R. L. Zusi. 2001. Higher-order phylogenetics of modern Aves based on comparative anatomy. Netherlands Journal of Zoology 51:179-205.
Mayr, G., and J. Clarke. 2003. The deep divergences of neornithine birds: a phylogenetic analysis of morphological characters. Cladistics 19:527-553.
Parker, T.J. 1891. Observations on the anatomy and development of Apteryx. Philosophical Transactions of the Royal Society of London, B 182: 25-134
Parker, W.K. 1866. On the structure and development of the skull in the ostrich tribe. Philosophical Transactions of the Royal Society of London 156:113-183.
Proctor, N.S. and P.J. Lynch. 1993. Manual of Ornithology. Yale University Press.
Pycraft, W. P. 1900. On the morphology and phylogeny of the Palaeognathae (Ratitae and Crypturi) and Neognathae (Carinatae). Transactions of the Zoological Society of London 15:149-290.
Saiff, E. I. 1981. The middle ear of the skull of birds: Struthio camelus. Zoological Journal of the Linnean Society, London 73:201-212.
Sauer, E. G. F., and E. M. Sauer. 1966. The behaviour and ecology of the South African ostrich. Living Bird 5:45-75.
Webb, M. 1957. The ontogeny of the cranial bones, cranial peripheral and cranial parasympathetic nerves, together with a study of the visceral muscles of Struthio. Acta Zoologica 38: 81-203.
Links
Struthio camelus on the Animal Diversity Web (The University of Michigan Museum of Zoology)
The Oklahoma State University School of Veterinary Medicine Ostrich Book
Hungry? Try The Ostrich Growers' Meat Company
Feel like cooking it yourself? Get ten free ostrich meat recipes at Ostriches Online
And, if you're going hiking or camping, you can't go wrong with the House of Jerky

Literature
& Links
None available.

Additional
Imagery
|

