Cynopterus brachyotis, the lesser short-nosed fruit bat, is a member of Pteropodidae, frequently called megachiropteran bats. There are two main groups of bats: megachiropterans (Old World fruit bats) and microchiropterans (echolocating bats:
Eptesicus fuscus,
Hipposideros gigas,
Carollia perspicillata).
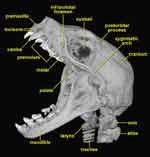 |
Skull of Cynopterus brachyotis. |
|
All megachiropterans are nectar and pollen feeding or frugivorous with long heads and jaws with large eyes, an unspecialized nose pad, and no tragus (a cartilaginous fold at the notch of the ear). Cynopterus brachyotis is native to India and southeast Asia, where it feeds on fruits and flowers. Megachiropterans do not echolocate, but orient instead using their large eyes. Their vision is very good even in near darkness, although they have a more direct steady style of flight compared to the more maneuverable microchiropterans.
|
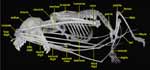 |
Lateral view of Cynopterus brachyotis. |
|
Most megachiropterans have claws on the first and second digits of their hand, which they use while moving around in trees. Microchiropterans, which do not tend to roost in trees, lack the second digit claw. Many megachiropterans lack tails completely, although some (like this species) have short ones; these are never ensheathed in the uropatagium (membrane between hind legs). They include the largest bats at about a kilogram in weight and a wingspan of more than a meter, however, most are small to medium sized. |
Bats are cosmopolitan in distribution with the majority found in the tropics. There are more bat species in the tropics than all other mammal species combined. They are thought to have descended from a common ancestor with colugos (Dermoptera), and are more distantly related to tree shrews (Scandentia) and primates (). The bat fossil record dates back to the early Eocene by which time they were already highly specialized. Today, bats play a major roll in terrestrial ecosystems as pollinators, seed dispersers, and predators of insects. Plants that have adapted to use bats as pollinators usually have downward facing white, funnel-shaped flowers with a musky odor, ensuring the pollen sticks to the bat's face.
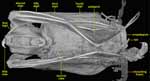 |
Ventral view of Cynopterus brachyotis. |
|
Bats are the only mammals to have evolved powered flight, as evidenced by their highly modified skeletons. Unlike birds, who have feathered wings that are supported by the first and second digits, bats have a thin membrane (patagium) that covers digits two through five and attaches to the side or back of the body and the leg. Another membrane (uropatagium) stretches between the hind legs; the tail may or may not be incorporated into the uropatagium. The first digit (thumb) is clawed and free, functioning in grasping and manipulation as in some other mammals. |
The bones of the wing are the same as those in the forelimbs of all tetrapods, although the proportions have been modified for active flight. Elbow and wrist movement is limited to prevent rotation of the joints, eliminating the need for musculature to control such movement. Additionally, the extension and flexion of the wing is accomplished using muscles closer to the body to reduce the need for forearm and wrist musculature. This reduction in musculature reduces the mass (and therefore kinetic energy) of the distal portion of the wing, resulting in greater control.
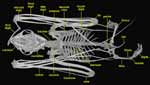 |
Ventral view of Cynopterus brachyotis. |
|
The humerus and radius are also reduced in thickness, similar to bird bones, with the cortical bone reduced to deal with the torsional stresses generated during flight. In bats, flying uses many muscles in the back during the upstroke and in the back and chest during the downstroke. In birds, the upstroke and downstroke are powered by chest muscles, resulting in reinforcement of the sternum and ribs. Because bats use many different muscles for flight, only one of which originates on the sternum, they do not have a reinforced sternum. The scapula is braced only against the clavicle, permitting it to rotate on its long axis; this enables bats to use a broader configuration of muscles for flight. |
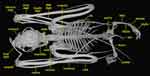 | Dorsal view of Cynopterus brachyotis. |
| Another adaptation of bats is the rotation of the hip joint 90 degrees (or 180 degrees in a few species), making the legs turn outward. This rotation allows them to roost hanging upside down by their legs, although it does make their gait on ground awkward. Roosting changes the usual forces on the legs from compressional stresses, as with most mammals, to tensional stresses. To accommodate this, tendons in the toes lock the claws together to reduce the muscular energy required to grasp and allow for sleep.
|
Bats roost in caves, rock crevices, trees, or man-made structures either solitarily or in colonies ranging in size to over a million individuals. Roosting is beneficial as a means of quick takeoff when threatened. Social bats that live in colonies have complex social behaviors, including polygamy and vocal communication signals, and even reciprocal altruism has been observed in some species.

About the Species
This specimen was collected in Malaysia prior to 1970. It is now part of the American Museum of Natural History Mammalogy Collections (AMNH 232506). The specimen was made available for scanning by Dr. Nancy Simmons of the American Museum of Natural History. Funding for scanning was provided by a National Science Foundation grant (DEB-9873663) to Dr. Simmons, and funding for image processing was provided by a National Science Foundation Digital Libraries Initiative grant to Dr. Timothy Rowe of the Department of Geological Sciences, The University of Texas at Austin.

About this Specimen
The specimen was scanned by Matthew Colbert on 8 May 2002 along the coronal axis for a total of 242 slices, each slice 0.0249 mm thick with an interslice spacing of 0.0195 mm. 
About the
Scan
Literature
& Links
Animations of the carpals around the three orthogonal axes: |
 |
 |
 |
Roll (1.1mb) |
Pitch (1.2mb) |
Yaw (1mb) |
|
Animations of the digits around the three orthogonal axes: |
 |
 |
 |
Roll (1.8mb) |
Pitch (2.4mb) |
Yaw (1.8mb) |
|
Animations of the radius around the three orthogonal axes: |
 |
 |
 |
Roll (1.4mb) |
Pitch (1.6mb) |
Yaw (1.4mb) |
|
Animations of the scl around the three orthogonal axes: |
 |
 |
 |
Roll (1mb) |
Pitch (1mb) |
Yaw (1mb) |

Additional Imagery
 |

